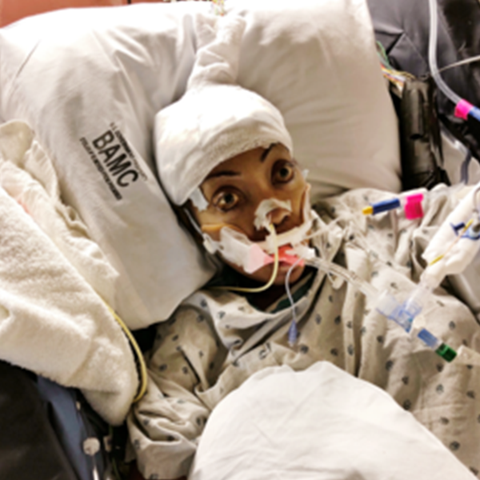
ANTWAN PIPER WAS at dinner with colleagues on a work trip in Oklahoma when his wife, Alexis, called from back home in Texas. He figured he would call her back later, but then she called again. On the third call, he finally picked up. Alexis sounded out of breath and in pain, her voice tight. “I’ve already called the ambulance,” she managed to get out.
It was Sunday, March 4, 2018, and Alexis was in “crisis”—a term for when her sickle-cell anemia flares up. Due to this congenital condition, Alexis’s red blood cells—normally round and flexible—become rigid and sticky, and twist into a sickle-like shape, rendering them poor at carrying oxygen, job number one for red blood cells. They clump together and can get stuck in small blood vessels, blocking blood flow and oxygen in certain parts of the body, which causes excruciating pain.
Except this time felt different, far worse than ever before. In the span of several hours, Alexis had gone from feeling like she had a cold to experiencing shooting pain throughout her body, leaving her crumpled on the floor of their upstairs hallway. Antwan served as a master sergeant in the Air Force and a church pastor, while his wife, a former teacher, wrote upbeat and inspirational religious books.
Both were generally healthy and in their mid-30s, and they always tried to stay positive. At the moment, however, both were terrified. “I don’t think words can do it justice, the feeling of a husband hearing his wife in pain and he can’t do anything about it,” Antwan says.
When Alexis arrived at Brooke Army Medical Center, outside San Antonio, the doctors quickly went through the usual protocols for a sickle-cell patient in crisis, ultimately electing to give her a transfusion. They checked her blood type, found a match, and tapped a fresh bag of red blood cells into her IV, hoping they would diffuse among the damaged ones. After a few days of additional treatment, her doctors decided to give her another transfusion to try to raise her level of hemoglobin, a blood protein that transports oxygen. During this second transfusion, Alexis began writhing and screaming. The pain was unbearable—“a ten out of ten,” she says.
Alexis’s hematologist, Lauren Lee, M.D., recognized the problem immediately: The patient had been receiving transfusions since age seven. Now she’d entered a potentially fatal state of hyperhemolysis, a condition in which the immune system attacks red blood cells, believing them to be an invading pathogen, which further hampers fresh oxygen transportation to strangled cells.
A healthy person’s hemoglobin level is 12 to 15 grams per deciliter. Because of her condition, Alexis’s typical level was 7, but it had dropped to about 5 by the time she was admitted. It kept falling as she slipped in and out of consciousness and hospital staff rushed her to the ICU.
By the time Antwan reached her bedside, things looked bleak. Alexis’s hemoglobin level had dropped into the 2-to-3 range, and she was very weak. “That’s when they started prompting me, preparing me for her dying,” he says. “They’re telling me there’s been no survivors” among patients with such a low hemoglobin level.
But Dr. Lee had one last idea. She got in touch with two top hematologists—Marc Zumberg, M.D., and Colonel Andrew Cap, M.D., Ph.D.—and everyone thought one risky intervention might still be worth a shot. It was a transfusion of, well, not blood exactly but rather an approximation called Hemopure, a synthetic blood-like product made from cow blood.
Because Hemopure is not actual blood, it could theoretically be given to just about anyone. It’s shelf stable and adaptive to any blood type. Gin up enough and you could have it on tap in ambulances and Army-medic backpacks, just about anywhere you might need a lifesaving transfusion on demand.
The catch: Hemopure has not yet been approved by the FDA. In the U. S. it’s only available through “compassionate use,” for patients with life-threatening conditions who have exhausted all other treatment options. Dr. Lee said there was no guarantee that it would work, and no guarantee that it would be free of side effects. All it had to do was deliver oxygen throughout Alexis’s body until she was well enough to start making her own blood cells. Dr. Lee asked Antwan if he was interested, and he said he wanted to pray over it. “It’s either this or she dies,” she remembers saying. “We wanted to give it a shot.”
Antwan agonized over all of these thoughts as he prayed. “They told me what they were hoping it would do, but they couldn’t guarantee me that it would work,” he says. “We were exhausted, and we didn’t have time. I really struggled, because of the uncertainty and the side effects they really didn’t know.” He couldn’t ask Alexis since she was unresponsive. It was all up to him.
LATE ONE NIGHT, Zaf Zafirelis’s phone started beeping and buzzing. He picked it up, knowing immediately what kind of call this would be: another desperate surgeon inquiring about Hemopure, the blood substitute made by the company he ran. Zafirelis was CEO of a tiny biotech outfit called Hemoglobin Oxygen Therapeutics, which had been operating on a shoestring but had recently secured space to open a new manufacturing facility across the street from a slaughterhouse in Pennsylvania.
He was used to getting three or four calls a week, always about patients whom conventional interventions had failed, always in the direst of straits. One time, it was a young man on Long Island with a rectal hemorrhage. Another, a 28-year-old woman in Michigan who needed risky heart surgery. Then there was the 35-year-old patient with anemia as a result of her leukemia. And the man in his 70s with a potentially fatal GI bleed.
Sometimes these calls came from doctors with patients who had severe anemia and no options, like Alexis. Often they involved patients who could not receive blood transfusions because of their religious beliefs, like Jehovah’s Witnesses, whose interpretation of the Bible prohibits “ingesting” the blood of their fellow man.
After a short talk with Dr. Lee, Zafirelis shipped ten units of Hemopure via emergency medical courier straight to San Antonio. He knew from experience that it could give her a shot. “The product will keep a patient who needs oxygen alive almost for as long as needed,” he says somewhat carefully.
Hemopure and products like it first came about as a response to the HIV/AIDS crisis of the 1980s, when many patients were unwittingly infected with the virus via blood transfusions. That led to a rush by several companies to transcend science fiction with a safe, pathogen-free alternative to natural blood.
The problems with traditional blood supplies are fairly straightforward. You can run out, donations can be tainted with pathogens, and the stuff doesn’t last that long outside the body—even chilled, you’ve got just a few weeks before packed red blood cells lose effectiveness.
Beginning in the late ’80s, scientists set out to come up with a replacement that would be not only disease-free but also stable without refrigeration. Whoever invented it certainly stood to prosper. “That was like the Holy Grail of blood,” says Kendall Crookston, M.D., Ph.D., a professor of medicine at the University of New Mexico School of Medicine. “But they pushed it from the bench into clinical trials perhaps too quickly.”
Two early formulations emerged, both focused on doing blood’s main job of transporting oxygen from the lungs to cells. The first was a red and syrupy class of artificial hemoglobin-based oxygen carriers (HBOCs) that replicated the action of red blood cells. The second was a milky set of compounds called perfluorocarbons, which transported oxygen chemically. By the early aughts, there were more than half a dozen products in various stages of development. But during clinical trials for two products, PolyHeme (an HBOC) and Oxygent (a perfluorocarbon), people died and trials were halted. Investors fled, and the sector promptly tanked.
Hemopure survived, but barely. Unlike the other HBOCs, it was made from cow blood and not expired human blood. It contained no actual red or white blood cells, plasma, lipoproteins, or any other ingredient of the complex cocktail that is human blood. Instead, it delivered oxygen via hemoglobin made by separating the red blood cells out of cow blood, then lysing or chemically piercing them to produce microscopic hemoglobin molecules. Further (and proprietary) processing and polymerization rendered the hemoglobin into a solution easily processed by the body.
Hemopure lacked many of the properties of whole blood—it didn’t clot or create new antibodies. Could it keep you from bleeding out? Maybe, so long as your body caught up and eventually took over the healing process. But Zafirelis, who helped pioneer Hemopure at a company called Biopure, didn’t like how market speculation seemed to gloss over the drawbacks. He left the company in 1995. “It’s an unreasonable comparison,” he says. “There’s no way an artificial product can match what the real thing does.”
The SEC alleged that, starting in 2003, Biopure had failed to publicly disclose that the FDA had put a hold on a clinical trial of the product in emergency-room trauma patients due to safety concerns. The company’s stock crashed, and three executives were indicted for making false representations about the health of the venture. Zafirelis was brought back as CEO in 2004 and helped popularize a veterinary HBOC version that has saved dogs, tropical parrots, an African rhino (through penile infusion), even a shark at a Vegas casino.
But the worst was yet to come. In April 2008, The Journal of the American Medical Associationpublished a meta-analysis of clinical trials of five HBOC products, including Hemopure, partially funded by the Washington, D. C., public-interest group Public Citizen. The authors added up the fatalities in the treatment and control groups from hundreds of combined treatments and concluded that these products actually increased a patient’s risk of heart attack and possibly death because they “scavenge” nitric oxide, a blood-borne gas that causes blood vessels to expand. Without it, blood vessels constrict. That’s a danger, but so is dying while doing nothing.
In 2009, the venture was sold to a new set of investors, who kept Zafirelis on board to see if they could resurrect the dream. They downsized dramatically and were trying to figure out how to reboot. Zafirelis’s company was on life support.
THE TEN UNITS of Hemopure reached the hospital less than 24 hours after Dr. Lee called Zafirelis. Unlike blood, which is shipped in ice-packed coolers, it arrived in a simple cardboard box. Each plastic bag inside sloshed with 250 milliliters of a dark-red liquid whose color reminds Zafirelis more of cabernet sauvignon than of actual blood.
Brooke Army Medical Center’s care team hitched the bag to Alexis’s IV, and its contents began to flow in. Whereas blood can take some time to start working, artificial hemoglobin transports oxygen quickly. And because the molecules are so tiny, about one-thousandth the size of red blood cells, they could easily filter into Alexis’s partially blocked blood vessels. Dr. Lee and the rest of the team watched as her hemoglobin level stabilized and then slowly began to climb.
Meanwhile, Dr. Lee gave Alexis other drugs and vitamins that she hoped would help her body begin to produce its own red blood cells again, as well as another experimental drug to stop the inflammatory reaction that was breaking down her existing red blood cells.
Over the next day or so, they infused another unit of Hemopure, and another, and another. Alexis woke up. “By about the fourth bag, she was able to have a conversation,” Dr. Lee recalls. “She looked the best she had ever looked.”
When the fifth bag was tapped, all that changed instantly. Alexis’s eyes fluttered and she started violently seizing, not just once but over and over. Dr. Lee wondered if Alexis was receiving too much oxygen, so she stopped the treatment and the seizures halted. Alexis’s hemoglobin level was dropping again.
Antwan had to face the same question with even higher stakes: Keep trying or let her go? Would he allow them to continue the Hemopure treatment on his wife, even if it risked causing more seizures—and possibly brain damage?
ZAFIRELIS HAS ZERO uncertainty about why he continues to pursue the dream of substitute blood, and why it matters. He thinks it’s time to drop comparisons and reset expectations completely. “The mistake that previous companies made was that they started claiming this was ‘artificial blood,’ which it’s not—it’s red cells only,” Zafirelis says. “This is where the industry hype ran away from science.” (So much so that in 2007, Danish cyclist Michael Rasmussen was forced to quit the Tour de France while in the leader’s yellow jersey because he’d somehow secured Hemopure as a performance enhancer. “It was useless,” he later tweeted. “Pure placebo.”)
Zafirelis’s plan is to test Hemopure in situations in which real blood simply doesn’t make sense, by working with the military (which receives FDA priority for the development of lifesaving medical products) and patients who have no alternative left. The Department of Defense recently committed $8.2 million to study Hemopure in up to 1,600 “pre-hospital” trauma patients in South Africa, where ambulance-transport times are far longer than the 11-minute average in the U. S. and clean blood is in short supply.
In fact, Hemopure has been approved for hospital use in South Africa for decades. “It has a very good safety record over 20 years, but we don’t have a lot of data for its use in trauma situations, which is why we want to do this study,” says Lee Wallis, M.D., the head of the division of emergency medicine at both the University of Cape Town and Stellenbosch University, who is in charge of the project.
Bruce Spiess, M.D., a professor of anesthesiology at the University of Florida, points out the broader potential for doctors in emergency situations throughout the developing world, where blood is sparse and diseases are harder to control.
Zafirelis is also working with a consortium of U. S. medical institutions, including the University of Michigan, Duke University, the University of Texas Southwest Medical Center, and the University of Rochester, to look at future applications of products like Hemopure. That’s part of a $4.8 million National Institutes of Health study examining the overall impact of those last-ditch interventions, even if they fail. “It’s often a choice between giving patients something and giving them nothing,” says Dr. Crookston.
But in the eyes of the FDA, the notion of substitute blood is a hypothesis that still needs testing—and some patients are in the unenviable position of being able to help with that. Their unfortunate health issues offer opportunities for doctors to learn new ways of assisting others. To not take advantage of that prolongs the inability to help those in similar situations. That’s part of why Zafirelis still answers his phone every time, but also why he’s encouraging less invasive experiments with just as much potential.
Hemopure stands to revolutionize not just critical and emergency care but transplants, too. In 2014, for instance, a Dutch transplant surgeon named Robert Porte requested Hemopure for a study testing how a perfusion system that circulates fluids might recondition 16 donated livers that had been initially declined for regular transplantation. After the treatment, 11 were deemed viable and were successfully transplanted into patients who had no other option for treating their advanced liver disease. These patients were severely ill at the time, but all 11 of them (and their livers) were doing fine six months later.
Normally, transplant surgeons weigh the risks and make a call: Is this organ healthy enough to help the recipient survive? “There is no way you can do a test drive,” Dr. Porte says. “You can’t take a risk with a patient’s life, so you have to err on the safe side. At the same time, people are dying on the wait list because we don’t have enough good organs.”
If Dr. Porte’s findings pan out, he estimates that the technique could increase the supply of available livers by 20 percent. In the meantime, Wake Forest University and the Cleveland Clinic are studying the implications for hand and limb transplants. And the journal Nature recently shared a study that shows perfusion with Hemopure can revive dead pig-brain cells, something that might eventually help stroke victims.
Tests on live pigs reveal that Hemopure may also affect ischemia—in which blocked blood vessels may damage the heart or the brain—by bringing oxygen there more quickly and efficiently than real blood can. In each case, the promise of a cure-all is being more narrowly defined, strategically sharpened, and potentially firmed up for more investment. Through success and, yes, sometimes failure, Zafirelis has learned that Hemopure can deliver oxygen to tissues and keep them alive outside of the body, in some cases more effectively than natural blood. “You have to do it under the right conditions,” he says.
“WE DON’T KNOW what kind of condition she’ll be in,” Antwan remembers the doctors telling him. “We won’t know how much or the degree of brain damage she may have suffered until she wakes up or starts moving around.” He wanted to pray again. “There is no time to pray,” they told him. “You have to decide now.”
After talking to a friend from church, Antwan decided that Alexis’s best chance was to keep going. Alexis received a sixth unit of Hemopure, enough to sustain her until her body could begin producing its own red blood cells with their own hemoglobin. Shortly after the last dose drained in, she was placed in a medically induced coma to prevent further seizures and assist her body with recovery. But even after her condition stabilized and she woke up, she faced a long and difficult road.
Alexis spent six weeks total in the hospital, often feeling foggy and confused. When her hemoglobin climbed back to 5.5—around the same level as when she was admitted—she was discharged to keep recuperating at home.
The oxygen-deprived state had damaged her muscles and motor skills, so she spent several months in physical and cognitive therapy with a home nurse, learning to walk again. Antwan took time off from work to be with her as she recovered. He doesn’t regret his decision to use Hemopure, and Alexis doesn’t, either. She’s back working on her books and views the experience as nothing short of a miracle: “I’m grateful that God could use something that hadn’t been done before and allow it graciously to help me in a very tough time and an ugly, life-threatening situation.”
Zafirelis remains hopeful. He recently got a call from a 19-year-old male Jehovah’s Witness in North Carolina with sickle-cell disease, who received 23 units of Hemopure. Another came from a 28-year-old man in Nebraska with sickle-cell complications cascading into pneumonia, respiratory failure, and acute congestive heart failure. He received 27 units over the course of more than two weeks. Both survived. All told, more than $1 billion has been invested in research and development for Hemopure, and the product has saved more than 400 people in life-or-death situations. That survival rate should only increase with more chances.
“This has become my life’s work now,” Zafirelis says. “I cannot give up. I believe in it. I’ve seen it work.” He’s ready to pull more bags out of storage the next time the phone rings.
via What Is Hemopure? The Blood Substitute That Could Save Lives